Lecture 1 Video 9 Peptide Bond Summary
🧬 Proteins Are Built from Amino Acids
Proteins are polymers built from amino acids, and in biological proteins these are always L-amino acids. Each amino acid has:
- an amino group (–NH₂)
- a carboxyl group (–COOH)
- a Cα (alpha carbon) with a side chain (R-group)
🔗 Formation of the Peptide Bond
- The carboxyl group of one amino acid reacts with the amino group of another.
- This is a condensation reaction, releasing one molecule of water (H₂O).
- The resulting linkage is an amide bond, called a peptide bond.
📌 A chain of amino acids linked this way is called a peptide (or polypeptide for longer chains).
🔁 Directionality of Peptides: N → C

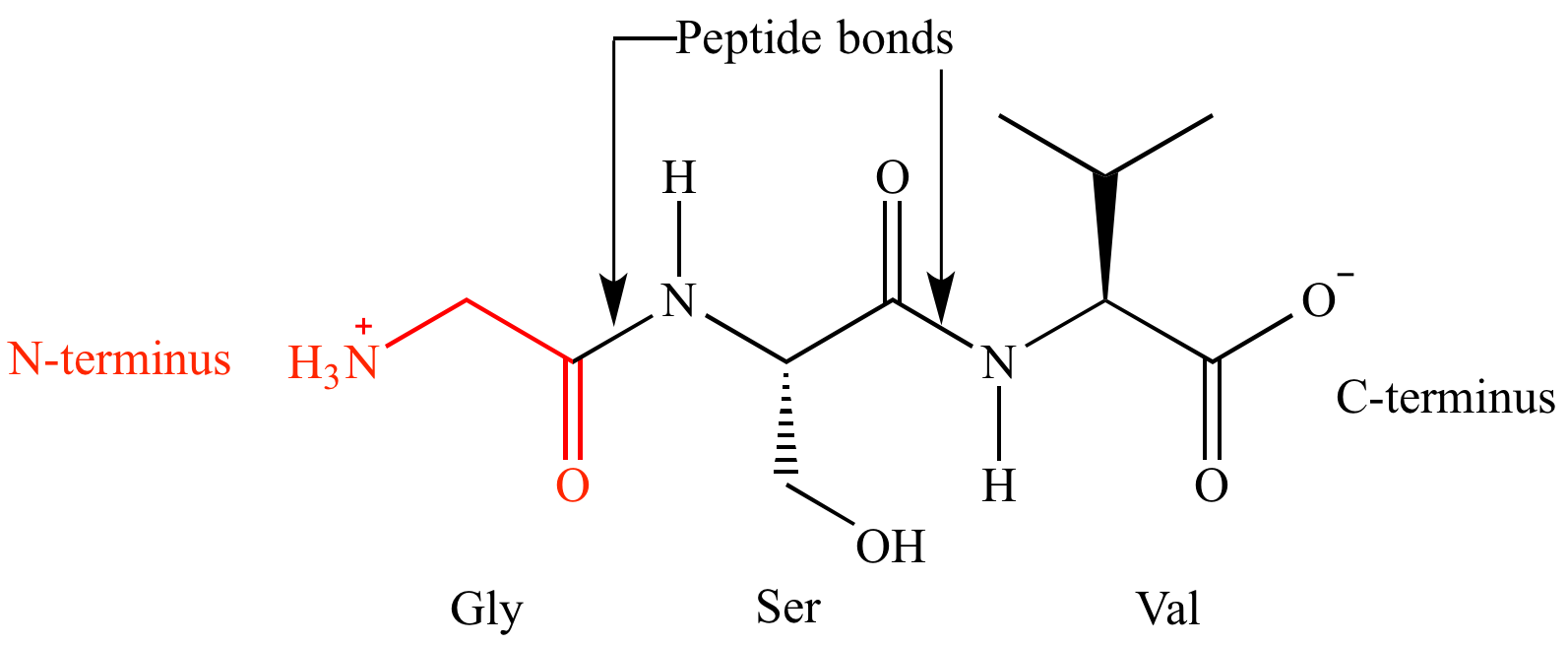
Peptides have directionality:
- N-terminus: free amino group
- C-terminus: free carboxyl group
➡️ Amino acid sequences are always written and read from N-terminus to C-terminus.
🧱 The Protein Backbone


The backbone is the repeating, invariant structure:
N - Cα - C
Side chains (R-groups) are attached to the Cα, but do not define the backbone geometry.
🔄 Conformation Depends on Rotation
Protein shape is controlled by rotations around backbone bonds. These rotations are described by dihedral angles, measured in degrees (°).
📐 What Is a Dihedral Angle?

A dihedral angle:
- Is defined by four atoms
- Describes rotation around the bond between the two middle atoms
- Requires viewing the molecule along that bond
🟦 Φ (Phi) Angle — Rotation Around N–Cα


- Bond rotated: Nᵢ – Cαᵢ
- Defined by atoms:
- C(i−1) → N(i) → Cα(i) → C(i)
How to visualize Φ:
- Look down the N–Cα bond
- N is in front, Cα hidden behind
- Measure the angle between:
- Cα → C(i)
- N → C(i−1)
🟨 Ψ (Psi) Angle — Rotation Around Cα–C


- Bond rotated: Cαᵢ – Cᵢ
- Defined by atoms:
- N(i) → Cα(i) → C(i) → N(i+1)
Same principle: look down the bond and measure relative atom positions.
📊 Common Dihedral Angle Values


In idealized staggered conformations, dihedral angles cluster around:
- −60° → gauche⁻ (G⁻)
- +60° → gauche⁺ (G⁺)
- 180° → trans (T)
📌 In real proteins, angles can vary—but are usually close to these values.
🔒 Ω (Omega) Angle — The Peptide Bond
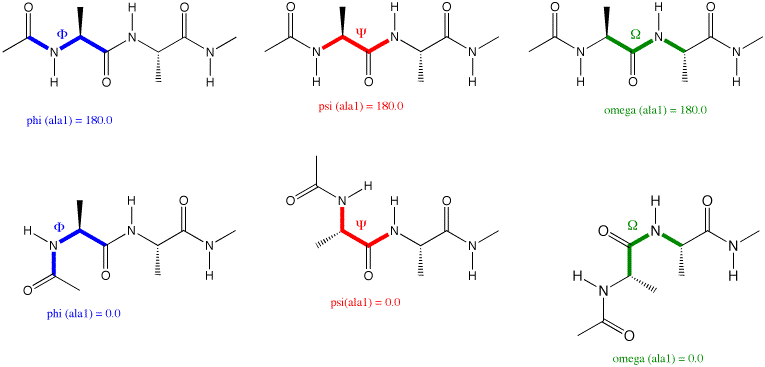
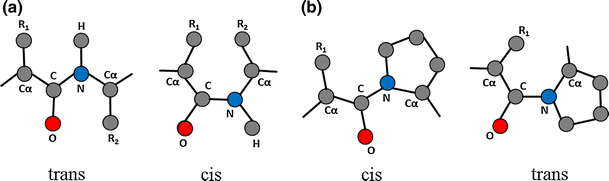
Ω describes rotation around the peptide bond (C–N).
Why Omega Is Special
- The peptide bond has partial double-bond character
- This makes it planar
- Rotation is highly restricted
✅ Only two values are allowed:
- 180° → trans
- 0° → cis
⚖️ Cis vs Trans Peptide Bonds


- Trans (ω = 180°)
- Vast majority of peptide bonds
- Minimizes steric clashes (atoms bumping into each other)
- Cis (ω = 0°)
- Rare
- Causes strong steric hindrance
📌 Exception: Proline
- Proline sometimes appears in cis
- Its cyclic side chain reduces the energetic penalty
🚫 Steric Hindrance (Why Cis Is Rare)
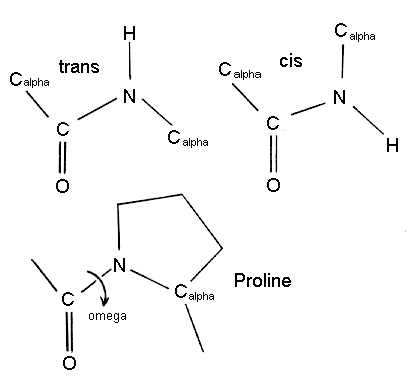
Steric hindrance means:
- Atoms occupy physical space
- Certain conformations cause atoms to overlap
- This raises energy and destabilizes the structure
➡️ Trans peptide bonds avoid these clashes, making them strongly favored.
🧠 Big Picture Summary
- Proteins are chains of L-amino acids linked by peptide bonds
- The backbone is N–Cα–C
- Conformation is controlled by three dihedral angles:
- Φ (phi): N–Cα
- Ψ (psi): Cα–C
- Ω (omega): peptide bond
- Φ and Ψ are flexible → determine secondary structure
- Ω is planar → almost always trans
- Proline is the notable cis exception


